Welcome to the Khalimonchuk Lab at the University of Nebraska
Work in our lab is focused on fundamental biological processes that involve mitochondria. To gain insights into vital aspects of mitochondrial biology and human disease and aging, we utilize several model systems and state-of-the-art genetics and biochemical approaches as well as various physiological and imaging techniques.
Department of Biochemistry
University of Nebraska
N230 Beadle Center
Lincoln, NE 68588-0664
402-472-8060
Research Overview:
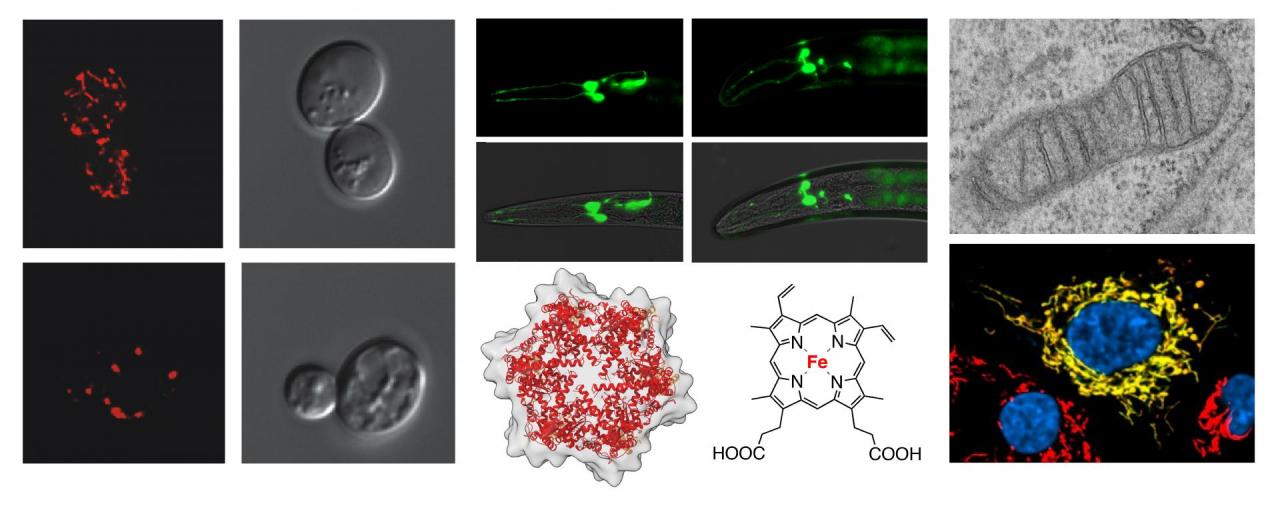
Mitochondria are complex and highly dynamic organelles responsible for a number of vital functions including cellular energy conversion, a plethora of metabolic and biosynthetic pathways, maintenance of ion homeostasis and cell death. Perturbations to mitochondrial function and integrity lead to dysfunctions that manifest in a spectrum of early- to adult-onset neurological and cardiovascular disorders, certain types of cancer, Type II diabetes and neurodegeneration. Understanding the molecular bases of mitochondrial function/dysfunction is a key to finding ways to combat these currently incurable diseases. Our research utilizes yeast, roundworm and mammalian cell culture models to address the following fundamental questions.
Role of protein quality control in mitochondrial homeostasis and stress responses
Mitochondrial respiration is inherently linked to generation of reactive oxygen species (ROS). In addition, redox-active intermediates in the biogenesis of the electron transfer chain respiratory complexes can further facilitate ROS production. Accumulating or persisting ROS can damage mitochondrial proteins and/or DNA located in the vicinity to respiratory chain, thereby contributing to mitochondrial dysfunction. Mitochondrial protein quality control (MPQC) system composed of molecular chaperones and proteases is a key factor that helps cells to cope with homeostatic challenges such as oxidative damage and protein misfolding. MPQC comprises a number of highly conserved proteases and molecular chaperones, important functions of which remain obscure. We seek to elucidate the individual roles of various MPQC components in preservation of mitochondrial functions and plasticity, and determine how impaired protein processing or turnover leads to aging-related diseases.
Mitochondrial heme transport
Heme is an essential, but inherently reactive and cytotoxic cofactor and signaling molecule. In most eukaryotes heme biosynthesis is initiated and completed within the mitochondria. All mitochondrial heme species are generated from heme b (aka protoheme) produced by the enzyme ferrochelatase and must be mobilized and trafficked for further distribution in virtually every subcellular compartment via largely unknown mechanisms. Our studies are aimed at understanding the mechanisms that govern safe mobilization, modification, transport and distribution of heme within and out of the mitochondria. Unraveling the molecular details of heme trafficking and export in health and disease states may identify targetable molecules for treatment of highly prevalent human disorders associated with heme and iron dyshomeostasis.
Biogenesis and maintenance of protein complexes within the inner mitochondrial membrane
The vast majority of proteins comprising mitochondrial proteome is synthesized in the cytosol and imported into the organelle, while only a handful of polypeptides originate from the mitochondrial genome. The inner mitochondrial membrane (IM) is an ultimate destination for many of these proteins wherein they are organized into high molecular weight complexes. Some of these assemblies such as cytochrome c oxidase (aka Complex IV), encompass proteins of dual genomic origin and harbor highly reactive prosthetic groups. To ensure normal mitochondrial function, numerous dedicated chaperones and assembly factors assist and regulate biogenesis and maintenance of these protein machineries. We seek to understand how the IM-localized multiprotein ensembles are formed and maintained, and how their erroneous biogenesis due to mutations in assembly factors drives clinical manifestations.
Oleh "Chuck" Khalimonchuk, Ph.D.
PhD, TU Dresden / Max Planck Insitute for Cell Biology & Genetics, Germany
Postdoctoral, University of Utah School of Medicine
Willa Cather Professor of Biochemistry

Research Interest:
I am interested in fundamental understanding of processes that underlie vital mitochondrial functions, and their contribution to complex maladies in humans.
Email Oleh
Iryna Bohovych, Ph.D.
PhD, University of Aberdeen, Scotland, UK
Senior Research Associate

Research Interest:
I am interested in functional crosstalk between proteases- and ubiquitin/proteasome-mediated mitochondrial quality control in yeast and human cells.
Email Iryna
Gunjan Purohit, Ph.D.
PhD, New Delhi Institute of Genomics & Integrative Biology, India
Postdoctoral Research Associate
Research Interest:
My research focus is to understand how failing mitochondrial fidelity contributes to aging and the development and progression of neurodegenerative diseases.
Email Gunjan
Nataliya Zahayko
MS, Lviv National University, Ukraine
Research Technician
Research Interest:
I am interested in developing genetic tools in the roundworm Caenorhabditis elegans model that will allow us to study how mitochondrial dysfuntion contributes to aging-related neuromuscular diseases in humans.
Email Nataliya
Alondra Torres-Gerena
BS, Interamerican University, Puerto Rico
Graduate Student

Research Interest:
I am interested in delineating molecular basis of redox regulation of mitochodnrial quality control factors to elucidate their role in mitochondrial stress remodeling and relevant disease states.
Email Alondra
Polash Ghosh
BS and MS, Mawlana Bhashani Science & Technology University, Bangladesh
Graduate Student
Research Interest:
I am interested in delineating the role of a conserved metalloprotease in the mitochondrial protein homeostasis and its contribution to human neuromuscular disorders.
Email Polash
Emma Bergmeyer
BS, University of Nebraska-Lincoln
Graduate Student

Research Interest:
I am interested in understanding the role of a novel mitochondrial AAA+ ATPase and its contribution to cellular protein homeostasis and aging.
Email Emma
Saieeda Fabia Ali
MS, Tzu Chi University, Taiwan
Graduate Student

Research Interest:
I am interested in understanding the mechanisms of mitochondrial heme transport and associated pathological states.
Email Fabia
PubMed Link
Complete list of our published work can be found at:
https://scholar.google.com/citations?user=k2OzIN0AAAAJ&hl=en&oi=ao
http://www.ncbi.nlm.nih.gov/myncbi/oleh.khalimonchuk.1/bibliography/public/
Selected Recent Publications:
1. Nyvltova, E., Dietz, J.V., Seravalli, J., Khalimonchuk, O., Barrientos, A. (2022) Coordination of metal center biogenesis in human cytochrome c oxidase. Nature Commun. 13, 3615.
2. Dietz, J.V., Bohovych, I., Willoughby, M.M., Piel 3rd, R.B., Ross, T.A., Addis, H.G., Fox, J.L., Lanzilotta, W.N., Dailey, H.A., Wohlschlegel, J.A., Reddi, A.R., Medlock, A.E., Khalimonchuk, O. (2021) Mitochondrial contact site and cristae organizing system (MICOS) machinery supports heme biosynthesis by enabling optimal performance of ferrochelatase. Redox Biol. 46, 102125.
3. Viana, M.P., Levytskyy, R.M., Anand, R., Reichert, A.S., Khalimonchuk, O. (2021) Protease OMA1 modulates mitochondrial bioenergetics and ultrastructure through dynamic association with MICOS complex iScience 24, 102119.
4. Martinez-Guzman, O., Willoughby M.M., Saini, A., Dietz, J.V., Bohovych, I., Medlock, A.E., Khalimonchuk, O., Reddi, A.R. (2020) Mitochondria-nuclear heme trafficking is regulated by GTPases that control mitochondrial dynamics and ER contact sites. J. Cell Sci. jcs.237917.
5. Daverey, A., Levytskyy, R.M., Swenson, S., Hayward, S.L., Stanke, K.M., Viana, M.P., Narasimhan, M., Khalimonchuk, O., Kidambi, S. (2019) Depletion of mitochondrial protease OMA1 alters proliferative properties and promotes metastatic growth of breast cancer cells. Sci. Rep. 9, 14746.
6. Bohovych, I., Dietz, J.V., Swenson, S., Zahayko, N., Khalimonchuk, O. (2019) Redox regulation of the mitochondrial quality control protease Oma1. Antioxid. Redox Signal. 31, 429-443.
7. Germany, E.M., Zahayko, N., Huebsch, M., Fox, J.L., Prahlad, V., Khalimonchuk, O. (2018) The AAA-ATPase Afg1 preserves organellar fidelity and cellular healthspan by maintaining mitochondrial matrix proteostasis. J. Cell Sci. 131, pii: jcs219956.
8. Tsushima, K., Bugger, H., Wende, A.R., Soto, J., Jenson, G.A., Tor, A.R., McGlauflin, R., Kenny, H.C., Zhang, Y., Souvenir, R., Hu, X.X., Sloan, C.L., Pereira, R.O., Lira, V.A., Spitzer, K.W., Sharp, T.L., Shoghi, K.I., Sparagna, G.C., Rog-Zielinska, E.A., Kohl, P., Khalimonchuk, O., Schaffer, J.E., Abel, E.D. (2018) Mitochondrial reactive oxygen species in lipotoxic hearts induce post-translational modifications of AKAP121, DRP1 and OPA1 that promote mitochondrial fission. Circ. Res. 122, 58-73.
9. Taylor, N.G., Swenson, S., Harris, N.J., Germany, E.M., Fox, J.L., Khalimonchuk, O. (2017) The assembly factor Pet117 couples heme a synthase activity to cytochrome oxidase assembly. J. Biol. Chem. 292, 1815-1825.
10. Bohovych, I., Kastora, S., Christianson, S., Kim, H.J., Fangman, T., Zhou, Y.J., Barrientos, A., Brown, A.J., Khalimonchuk, O. (2016) Oma1 links mitochondrial protein quality control and TOR signaling to modulate physiological plasticity and cellular stress responses. Mol. Cell. Biol. 36, 2300-2312.
Past Postdoctoral Fellows
Ganapathi Kandasamy, PhD, University of Cologne
Present position: Postdoctroal Researcher, University of Miami
Sang-Gyu Hwang, PhD, University of Ulsan College of Medicine
Present position: Research Scientist, University of Ulsan, South Korea
Roman Levytskyy, PhD, MBA, UCSD
Present position: Director of Scientific Communications & Publications, Nurix Therapeutics Inc.
Kacoli Sen Banerjee, PhD, Indian Institute of Technology
Present position: Research Associate, University of Florida
Past Graduate Students
Present position: Staff Scientist, Charles River Laboratories
Present position: Instructor in Statistics, University of Nebraska-Lincoln
Present position: Postdoctoral Fellow, Rutgers University
Present position: Senior Associate Scientist, Pfizer Inc.
Present position: Postdoctoral Fellow, University of Miyazaki, Japan
Present position: Instructor, University of Nebraska Medical Center
Present position: Research Associate, Doane University
Past Undergraduate Students
Applying to medical schools
MD student, University of Nebraska Medical Center
MPH student, University of Nebraska Medical Center
DDS student, Creighton University School of Dentistry
MD/PhD student, University of Utah School of Medicine
MD student, University of Nebraska Medical Center
MD, Anesthesiology Resident, Ohio State University Medical Center
Catholic Teacher Corps Program at Creighton University
DO, Resident Physician, CoxHealth Medical System
MD, Family Medicine Resident, Pella Regional Health Center
MD/PhD, Pathology Resident, Mayo Clinic